The Afrin Recall Nobody’s Explaining Correctly
Over 750,000 bottles of Afrin nasal spray have been yanked from shelves, and the headlines are calling it a “safety hazard” without telling you what that actually means for your nasal passages. Here’s what you need to understand: this isn’t about the medication itself—oxymetazoline works exactly as advertised—this is about microbial contamination that can turn your nasal cavity into a bacterial playground.
As a physician who’s treated countless sinus infections, I can tell you the media is burying the real story. The recall involves non-sterile manufacturing conditions, meaning bacteria, fungi, or other microorganisms could be living in those bottles. When you spray that into your nose, you’re not just delivering medicine—you’re potentially inoculating your nasal mucosa with pathogens.
Why Your Nasal Passages Are Uniquely Vulnerable
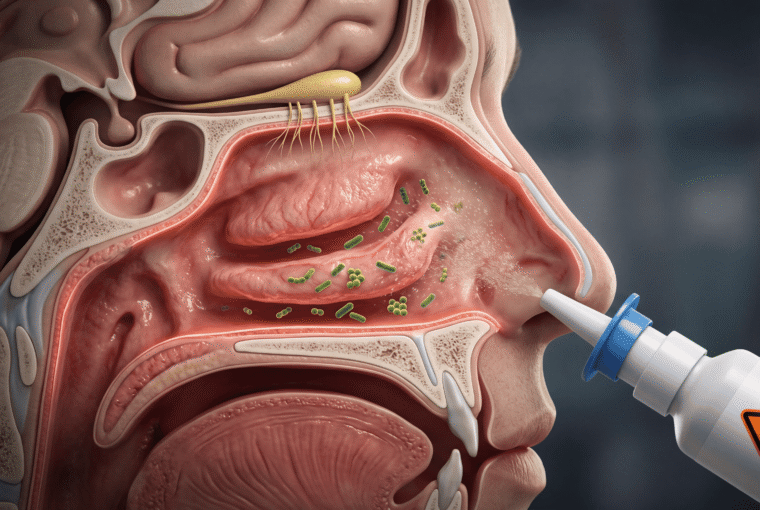
Your nasal mucosa isn’t like the skin on your arm. It’s a highly vascularized, permeable membrane specifically designed for rapid absorption—which is exactly why intranasal medications work so quickly. The flip side? Bacteria that contact this tissue can establish infection within hours, not days.
The nasal cavity maintains a delicate microbiome balance dominated by species like Staphylococcus epidermidis and Corynebacterium species that actually protect against pathogenic invaders. When you introduce contaminated solution, you’re disrupting this ecosystem. According to research published in the journal Microbiome, dysbiosis of the nasal microbiome is directly linked to chronic rhinosinusitis and increased susceptibility to respiratory infections.
Here’s the mechanism most doctors won’t take time to explain: contaminated nasal sprays can introduce Pseudomonas aeruginosa, Burkholderia cepacia complex, or even opportunistic fungi. These organisms thrive in the moist, warm environment of your nasal passages and can rapidly colonize damaged tissue—and yes, chronic Afrin use damages tissue through rebound congestion.
The Oxymetazoline Paradox: When Relief Becomes Risk
Oxymetazoline is an alpha-adrenergic agonist that constricts blood vessels in your nasal mucosa, reducing swelling and opening airways. It works brilliantly for 12 hours. But here’s what the box doesn’t emphasize enough: use it for more than three consecutive days, and you’ll trigger rhinitis medicamentosa—rebound congestion that’s often worse than your original problem.
The biological mechanism is straightforward but vicious. Prolonged vasoconstriction leads to compensatory vasodilation when the medication wears off. Your blood vessels become hypersensitive to normal circulating catecholamines, resulting in chronic swelling. Clinical studies in the American Journal of Rhinology show that rhinitis medicamentosa can persist for weeks after discontinuation, requiring intranasal corticosteroids to resolve.
Now layer microbial contamination onto already-damaged, chronically inflamed nasal tissue. You’ve created ideal conditions for infection. The compromised mucosal barrier can’t mount an effective immune response, and the organisms have direct access to submucosal tissues.
What The Media Got Wrong About This Recall
Every article I’ve seen focuses on the number of bottles recalled—750,000 sounds scary—but misses three critical points that actually matter to your health.
First, they’re not explaining that microbial contamination in nasal products is categorically different from contamination in topical creams. Your nasal mucosa connects directly to your sinuses, your middle ear via the Eustachian tube, and your lower respiratory tract. An infection that starts in your nose doesn’t stay there. I’ve seen patients develop sinusitis, otitis media, and even aspiration pneumonia from nasal-origin infections.
Second, the headlines aren’t distinguishing between bacterial and fungal contamination risks. Fungal contamination is particularly insidious because most people assume “infection” means bacteria. Aspergillus species can colonize the sinuses and are notoriously difficult to eradicate without systemic antifungals. According to data from the CDC, fungal sinusitis often requires surgical debridement in addition to medical therapy.
Third—and this frustrates me most—no one’s addressing the immunocompromised population. If you’re on chemotherapy, taking immunosuppressants for autoimmune disease, or living with HIV, a contaminated nasal spray isn’t just an inconvenience. It’s a potential route for disseminated infection. The nasal mucosa in immunocompromised patients lacks the robust innate immune response that healthy individuals rely on as their first line of defense.
The Manufacturing Failure You Deserve To Understand
The FDA’s recall classification matters here. This is a Class II recall, meaning “a situation in which use of or exposure to a violative product may cause temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is remote.” Translation: they’re betting most people won’t develop serious infections, but some definitely will.
Non-sterile manufacturing conditions can mean several things: inadequate water purification systems, contaminated equipment, improper environmental controls in the production facility, or failure in preservative systems. The FDA requires nasal sprays to meet stringent microbial limits because manufacturers recognized these risks decades ago.
Here’s what concerns me as a clinician: if quality control failures allowed contaminated product to reach distribution, how long was contaminated product on shelves before detection? Microbial contamination typically becomes detectable only after significant growth. That’s why most recalls happen after consumer complaints about foul odor, unusual appearance, or adverse events—not from routine testing catching a small deviation.
Why Your Symptoms Might Not Be “Just Allergies”
If you’ve used a recalled Afrin product, watch for these red flags that suggest established infection rather than simple rhinitis: purulent (yellow or green) nasal discharge, unilateral facial pain or pressure, fever, persistent post-nasal drip lasting more than 10 days, or worsening symptoms after initial improvement.
The timing matters. Bacterial colonization can occur within 24-48 hours of contaminated spray use, but clinical infection typically manifests after 3-7 days. Fungal infections have a more insidious onset, often presenting as chronic symptoms that patients dismiss as “allergies that won’t go away.”
Here’s the clinical pearl most primary care physicians miss: if you develop sinusitis within a week of starting a new nasal spray, the spray itself should be considered as a potential source, not just the condition you were treating. I’ve diagnosed several cases of iatrogenic sinusitis over my career, all linked to contaminated nasal products.
The Rebound Congestion Trap That Makes Everything Worse
Let’s address the elephant in the medicine cabinet: many people using Afrin are already trapped in the rebound congestion cycle. You use it because you can’t breathe. It wears off. Your nose swells worse than before. You use it again. Repeat indefinitely.
This chronic use pattern means you’re potentially exposing compromised nasal tissue to contaminated solution multiple times daily for weeks or months. Each application reintroduces organisms, prevents normal mucociliary clearance (your nose’s self-cleaning mechanism), and further damages the protective epithelial barrier.
The mechanism of rebound congestion involves downregulation of alpha-adrenergic receptors and structural changes in the nasal mucosa. Research in Clinical and Experimental Otorhinolaryngology demonstrates that chronic oxymetazoline use causes epithelial squamous metaplasia, loss of cilia, and inflammatory cell infiltration. This damaged tissue is precisely where introduced pathogens can establish infection most easily.
What You Should Actually Do Right Now
First, check your Afrin bottles immediately. The recall affects specific lot numbers—the FDA maintains an updated list on their website. Don’t assume your bottle is safe just because you bought it recently; distribution timelines mean contaminated product could have reached shelves months ago.
If you have a recalled product, stop using it immediately. Don’t finish the bottle to “not waste it.” The risk isn’t worth the convenience. Return it to the pharmacy for a refund or dispose of it properly—don’t pour it down the drain where it can affect aquatic ecosystems.
If you’ve been using a recalled product and develop any symptoms of infection, see a physician promptly. Don’t wait to “see if it gets better.” Mention specifically that you used a recalled nasal spray—this information changes the diagnostic approach and may prompt cultures that wouldn’t otherwise be ordered.
For breaking the Afrin cycle: transition to intranasal corticosteroids like fluticasone or mometasone. Yes, they take 3-7 days to reach full effectiveness. Yes, that week will be miserable. But it’s the only way to restore normal nasal function. Use saline irrigation to maintain moisture and clearance during the transition. Consider oral decongestants like pseudoephedrine for temporary relief, but avoid phenylephrine—it’s been proven ineffective at standard doses.
The Bigger Picture About Over-The-Counter Safety
This recall should shatter a dangerous assumption: that OTC medications are somehow less risky than prescription drugs. The FDA’s oversight of OTC products is real, but manufacturing standards vary significantly between companies. Generic manufacturers may use different facilities, different preservative systems, and different quality control processes than brand-name producers.
The nasal spray category is particularly vulnerable to contamination because these products are multidose containers with preservatives designed to prevent microbial growth during use. When manufacturing controls fail, the preservative system may be inadequate or absent, allowing organism proliferation before the product ever reaches your medicine cabinet.
According to FDA recall data, microbial contamination accounts for approximately 15% of all drug recalls annually, with nasal products and ophthalmic solutions representing disproportionately high-risk categories due to their direct access to sterile or semi-sterile body sites.
Why Prevention Beats Treatment Every Time
The best approach to nasal congestion isn’t reaching for a spray first. Start with saline irrigation using a neti pot or squeeze bottle with distilled or previously boiled water. This mechanically removes allergens and inflammatory mediators without chemical intervention.
For allergic rhinitis, intranasal corticosteroids should be first-line therapy, not last resort. They address the underlying inflammatory process rather than just masking symptoms. For infectious rhinitis, time and supportive care resolve most viral cases—decongestants only make you more comfortable during recovery, they don’t accelerate it.
Reserve oxymetazoline for specific situations: severe congestion preventing sleep, air travel with Eustachian tube dysfunction, or the first few days transitioning off chronic use. Use it intelligently, not reflexively.
Your Action Plan Moving Forward
Check your medicine cabinet today, not tomorrow. Verify lot numbers against the FDA recall list. If you have recalled product, document the lot number and return date for your records—this matters if you develop delayed complications.
Rethink your approach to nasal congestion. If you’re using any decongestant spray more than three consecutive days, you’re treating the symptom while creating the disease. Break the cycle now, because adding infection risk to rebound congestion is a combination your nasal passages can’t handle.
Understand that “over-the-counter” doesn’t mean “without risk”—every medication, every spray, every seemingly benign remedy carries potential for harm when manufacturing fails or when used outside evidence-based guidelines.
Your nasal passages are not a playground for preventable infections, and you deserve medications manufactured to the standards that your mucous membranes require.